Insulin Transduction Pathway – How Insulin Regulates Blood Sugar
The insulin transduction pathway represents one of the most critical cellular communication systems in the human body, directly controlling how cells respond to insulin and manage blood glucose levels. This complex molecular process begins when insulin binds to specific receptors on cell surfaces, triggering a cascade of intracellular events that ultimately allow glucose to enter cells for energy production. The pathway involves multiple proteins, enzymes, and signaling molecules working together to maintain proper blood sugar balance. When this system functions correctly, it prevents dangerous glucose fluctuations and supports overall metabolic health. Disruptions in the insulin transduction pathway can lead to insulin resistance, type 2 diabetes, and various metabolic disorders that affect millions worldwide.
Cellular Mechanisms Of The Insulin Transduction Pathway
The insulin transduction pathway operates through a sophisticated series of molecular events that transform an external insulin signal into meaningful cellular responses. When insulin molecules circulate through the bloodstream, they seek out target cells throughout the body, including muscle, fat, and liver tissues. These cells contain specialized insulin receptors embedded in their outer membranes, acting as molecular locks that only insulin can unlock.
The process begins with insulin binding to these receptors with remarkable specificity. Each insulin receptor consists of two alpha subunits and two beta subunits, forming a complex structure that spans the cell membrane. The alpha subunits remain outside the cell, serving as the binding site for insulin, while the beta subunits extend through the membrane and into the cell’s interior.
Once insulin attaches to the alpha subunits, it triggers a conformational change in the receptor structure. This change activates the beta subunits, which possess intrinsic tyrosine kinase activity. The activated receptor then undergoes autophosphorylation, adding phosphate groups to specific tyrosine residues within its own structure. This phosphorylation event serves as the initial spark that ignites the entire insulin transduction pathway cascade.
Key Components of Receptor Activation:
- Insulin receptor substrate proteins (IRS-1, IRS-2, IRS-3, IRS-4)
- Phosphoinositide 3-kinase (PI3K)
- Protein kinase B (Akt/PKB)
- Mammalian target of rapamycin (mTOR)
Ready to support your blood sugar management? Metformin offers proven glucose control with convenient home delivery from Better You Rx.
Signal Transmission Through Protein Cascades
Following receptor activation, the insulin transduction pathway moves into its signal transmission phase, where information travels through multiple protein layers within the cell. This phase demonstrates the remarkable efficiency of cellular communication systems, as a single insulin binding event can ultimately affect thousands of cellular processes simultaneously.
The phosphorylated insulin receptor serves as a docking station for various intracellular proteins, most notably the insulin receptor substrate (IRS) family of proteins. These IRS proteins act as molecular adapters, connecting the activated receptor to downstream signaling pathways. When IRS proteins bind to the phosphorylated receptor, they themselves become phosphorylated on multiple tyrosine residues, creating additional binding sites for other signaling molecules.
Phosphoinositide 3-kinase (PI3K) represents one of the most important proteins recruited to phosphorylated IRS proteins. PI3K consists of a regulatory subunit (p85) and a catalytic subunit (p110) that work together to modify membrane lipids. The enzyme converts phosphatidylinositol 4,5-bisphosphate (PIP2) to phosphatidylinositol 3,4,5-trisphosphate (PIP3), creating a critical signaling lipid that serves as a platform for further protein recruitment.
The generation of PIP3 attracts additional proteins containing pleckstrin homology (PH) domains, including protein kinase B (Akt) and phosphoinositide-dependent kinase-1 (PDK1). These proteins cluster at specific membrane locations where PIP3 concentrations are highest, facilitating their activation through proximity-induced phosphorylation events.
| Protein | Function | Location |
| IRS-1/2 | Adapter proteins | Cytoplasm |
| PI3K | Lipid kinase | Membrane |
| PDK1 | Serine/threonine kinase | Membrane |
| Akt/PKB | Central regulatory kinase | Cytoplasm |
| mTOR | Growth regulator | Cytoplasm |
Glucose Transport And Cellular Uptake Mechanisms
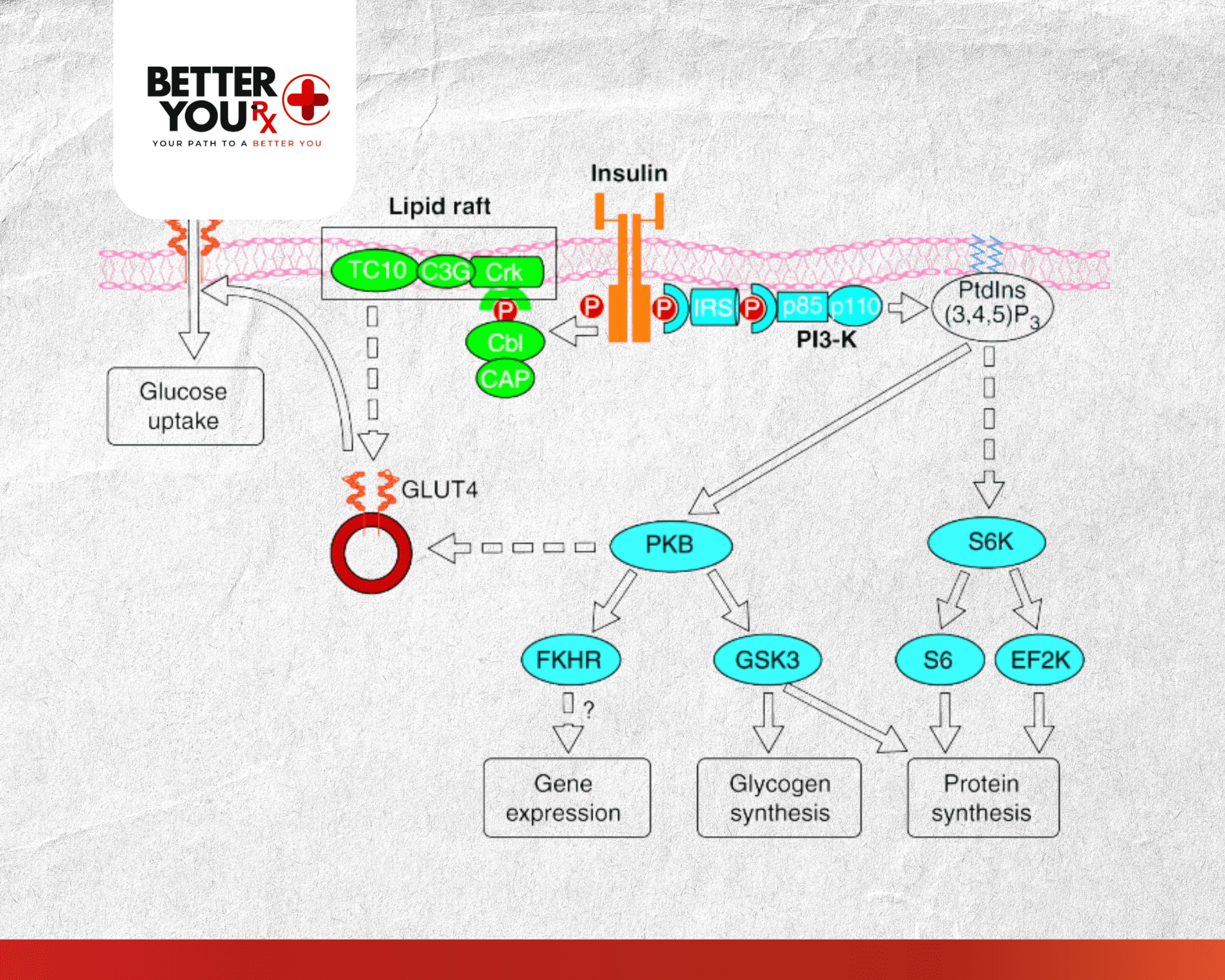
The insulin transduction pathway culminates in one of its most vital functions – facilitating glucose entry into cells where it can be utilized for energy production or stored for future use. This process involves the translocation and activation of specialized glucose transporter proteins that normally reside within intracellular vesicles.
Under basal conditions, most glucose transporter type 4 (GLUT4) proteins remain sequestered in intracellular storage vesicles, making cells relatively impermeable to glucose despite its abundance in the bloodstream. The insulin transduction pathway changes this situation dramatically by triggering the movement of these vesicles to the cell surface through a process called translocation.
Activated Akt plays a central role in GLUT4 translocation by phosphorylating and inactivating AS160 (Akt substrate of 160 kDa), a protein that normally prevents GLUT4 vesicle movement. When AS160 is phosphorylated and inactivated, it releases its inhibitory control over the vesicle trafficking machinery, allowing GLUT4-containing vesicles to move toward the cell membrane.
The vesicles carrying GLUT4 proteins fuse with the cell membrane through a complex process involving SNARE proteins and other membrane fusion machinery. Once incorporated into the membrane, GLUT4 transporters create pathways for glucose to enter the cell down its concentration gradient. This process can increase cellular glucose uptake by 10-20 fold compared to unstimulated conditions.
GLUT4 Translocation Process:
- Vesicle mobilization from intracellular pools
- Movement along cytoskeletal tracks
- Docking at the cell membrane
- Membrane fusion and GLUT4 insertion
- Glucose transport facilitation
Take control of your glucose levels today. Humalog provides rapid-acting insulin support available through Better You Rx with competitive pricing.
Metabolic Effects And Energy Storage Pathways
Beyond facilitating glucose uptake, the insulin transduction pathway orchestrates numerous metabolic changes that promote energy storage and utilization. These effects transform the cellular environment from a state optimized for energy mobilization to one focused on energy conservation and anabolic processes.
One of the primary metabolic effects involves glycogen synthesis in muscle and liver cells. The insulin transduction pathway activates glycogen synthase, the key enzyme responsible for converting glucose into glycogen for storage. This activation occurs through a multi-step process involving the inactivation of glycogen synthase kinase-3 (GSK-3) by Akt phosphorylation.
Simultaneously, the pathway promotes lipogenesis – the conversion of excess glucose into fatty acids for long-term energy storage. This process occurs primarily in liver and adipose tissue, where activated sterol regulatory element-binding protein-1c (SREBP-1c) increases the expression of lipogenic enzymes. The insulin transduction pathway also enhances the activity of acetyl-CoA carboxylase, the rate-limiting enzyme in fatty acid synthesis.
Protein synthesis receives significant enhancement through insulin signaling, particularly via the mTOR pathway. Activated Akt phosphorylates and activates mTOR complex 1 (mTORC1), which subsequently phosphorylates ribosomal protein S6 kinase 1 (S6K1) and eukaryotic translation initiation factor 4E-binding protein 1 (4E-BP1). These phosphorylation events promote ribosome biogenesis and translation initiation, increasing overall protein synthesis rates.
The pathway also inhibits gluconeogenesis – the production of glucose from non-carbohydrate sources. Akt phosphorylates and inactivates several transcription factors involved in gluconeogenic enzyme expression, including FOXO1 and PEPCK. This coordinated suppression of glucose production, combined with enhanced glucose uptake, helps maintain appropriate blood glucose levels.
Insulin Transduction Pathway Regulation And Feedback Mechanisms
The insulin transduction pathway incorporates multiple regulatory mechanisms that ensure appropriate cellular responses while preventing excessive activation that could lead to metabolic dysfunction. These regulatory systems operate at various levels, from receptor modification to downstream effector control.
Negative feedback regulation represents one of the most important control mechanisms within the pathway. Several proteins activated by insulin signaling subsequently dampen the signal through direct inhibition of upstream components. For example, S6K1, activated downstream in the pathway, phosphorylates IRS-1 on serine residues, reducing its ability to interact with the insulin receptor and effectively creating a negative feedback loop.
Protein tyrosine phosphatases (PTPs) provide another layer of regulation by removing phosphate groups from activated receptors and downstream proteins. PTP1B specifically targets the insulin receptor and IRS proteins, while PTEN dephosphorylates PIP3, converting it back to PIP2 and terminating PI3K-dependent signaling events. The balance between kinase and phosphatase activities determines the overall strength and duration of insulin signaling.
The pathway also responds to the nutritional state of the cell through various sensors and regulatory proteins. AMP-activated protein kinase (AMPK) serves as a cellular energy sensor that can modulate insulin sensitivity based on ATP availability. During energy stress, AMPK phosphorylates ACC and other metabolic enzymes, promoting catabolic processes while simultaneously enhancing insulin sensitivity to maximize glucose uptake.
Regulatory Mechanisms:
- Negative feedback loops
- Phosphatase-mediated signal termination
- Nutritional sensing systems
- Hormonal cross-talk
- Circadian rhythm influences
Pathological Disruptions In Insulin Transduction Pathway Function
When the insulin transduction pathway malfunctions, it can lead to serious metabolic consequences that affect millions of people worldwide. These disruptions occur at various points within the signaling cascade and can result from genetic factors, lifestyle influences, or combinations of both.
Insulin resistance represents the most common pathological disruption of the insulin transduction pathway. In this condition, cells become less responsive to insulin signals despite normal or elevated insulin levels in the bloodstream. The underlying mechanisms often involve chronic inflammation, which activates stress kinases like JNK and IKK that phosphorylate IRS proteins on serine residues, reducing their ability to transmit insulin signals effectively.
Chronic exposure to high glucose and fatty acid levels can also impair pathway function through several mechanisms. Hyperglycemia increases the production of advanced glycation end products (AGEs) that interfere with receptor function, while elevated free fatty acids activate protein kinase C isoforms that disrupt normal insulin signaling. These metabolic stresses create a vicious cycle where impaired insulin signaling leads to further metabolic dysfunction.
Genetic variations in pathway components can predispose individuals to insulin resistance and diabetes. Polymorphisms in genes encoding IRS proteins, PI3K subunits, or glucose transporters can alter protein function and reduce pathway efficiency. Additionally, mutations affecting receptor structure or expression can severely compromise cellular insulin sensitivity.
Oxidative stress represents another significant factor in pathway disruption. Reactive oxygen species generated during normal metabolism or pathological conditions can damage proteins within the signaling cascade, particularly at cysteine residues critical for protein function. This oxidative damage accumulates over time and contributes to age-related insulin resistance.
Support your metabolic health with proven solutions. Lantus provides long-acting insulin control available through Better You Rx at affordable prices.
Therapeutic Approaches And Treatment Strategies
Modern medicine has developed numerous therapeutic approaches that target various components of the insulin transduction pathway to restore normal glucose homeostasis and prevent diabetic complications. These treatments range from lifestyle interventions to sophisticated pharmaceutical agents that modulate specific pathway components.
Lifestyle modifications remain the cornerstone of therapy for insulin resistance and early diabetes. Regular physical exercise enhances insulin sensitivity through multiple mechanisms, including increased GLUT4 expression, improved mitochondrial function, and reduced chronic inflammation. Exercise also promotes the translocation of GLUT4 to the cell membrane through insulin-independent pathways, providing additional routes for glucose uptake.
Dietary interventions play crucial roles in optimizing insulin transduction pathway function. Caloric restriction and weight loss can reverse many of the metabolic abnormalities associated with insulin resistance, including reduced inflammation and improved cellular insulin sensitivity. Specific nutrients like omega-3 fatty acids and antioxidants may provide additional benefits by reducing oxidative stress and supporting healthy membrane composition.
Pharmacological treatments target different aspects of the pathway to enhance its function or compensate for its deficiencies. Some medications work by increasing insulin sensitivity at the receptor level, while others enhance downstream signaling events or provide alternative pathways for glucose disposal. The choice of treatment depends on the specific nature of the pathway disruption and individual patient characteristics.
Emerging therapeutic approaches focus on novel targets within the insulin transduction pathway. Research into small molecule activators of specific kinases, selective phosphatase inhibitors, and innovative delivery systems for pathway modulators continues to expand treatment options. These developments offer hope for more precise and effective interventions in the future.
Treatment Categories:
- Lifestyle interventions (diet and exercise)
- Insulin sensitizing medications
- Direct insulin replacement therapy
- Combination therapeutic approaches
- Novel molecular targets under development
Key Takeaways
The insulin transduction pathway represents a fundamental biological process that maintains glucose homeostasis and supports cellular energy metabolism. Through its complex network of molecular interactions, this pathway translates insulin signals into meaningful cellular responses that affect glucose uptake, energy storage, and metabolic regulation. When functioning properly, the insulin transduction pathway ensures stable blood glucose levels and prevents the metabolic complications associated with diabetes.
Disruptions in this pathway can lead to serious health consequences, but understanding its mechanisms provides valuable insights for developing effective therapeutic strategies. From lifestyle modifications that enhance pathway sensitivity to targeted medications that address specific molecular defects, modern medicine offers numerous approaches for optimizing insulin transduction pathway function and maintaining metabolic health throughout life.
Frequently Asked Questions
What Are The Steps Of Insulin Transduction Pathway?
The insulin transduction pathway follows a sequential process beginning with insulin binding to cell surface receptors. The activated receptor phosphorylates itself and recruits IRS proteins, which then activate PI3K to produce PIP3. This lipid messenger recruits and activates Akt, which phosphorylates numerous downstream targets to promote glucose uptake, glycogen synthesis, and other metabolic processes.
What Are The 4 Stages Of The Signal Transduction Pathway?
Signal transduction pathways generally consist of four main stages – signal reception, signal transduction, signal amplification, and cellular response. In the insulin transduction pathway, reception occurs at the insulin receptor, transduction involves the protein kinase cascades, amplification happens through multiple phosphorylation events, and the response includes glucose transport and metabolic changes.
What Is The Insulin Action Signaling Pathway?
The insulin action signaling pathway describes how insulin binding to receptors triggers intracellular changes that regulate glucose metabolism. This pathway primarily operates through the PI3K-Akt signaling axis, which controls glucose transporter translocation, glycogen synthesis, lipogenesis, and protein synthesis while inhibiting gluconeogenesis and glycogenolysis.
What Is The Pathway Of Transduction?
Transduction pathways convert external signals into intracellular responses through molecular cascades. In insulin signaling, transduction begins with receptor activation and proceeds through IRS protein phosphorylation, PI3K activation, PIP3 production, and Akt activation. Each step amplifies the signal and activates additional downstream effectors that ultimately modify cellular behavior.
What Are The Three Types Of Transduction?
The three main types of signal transduction include mechanical transduction (converting physical forces into signals), chemical transduction (converting chemical signals like hormones into cellular responses), and electrical transduction (converting electrical signals into biological responses). The insulin transduction pathway represents chemical transduction, converting the chemical signal of insulin into metabolic changes.
What Are The Steps Of The Transduction Process?
The transduction process involves signal recognition, receptor activation, intracellular signal propagation, signal amplification, and target activation. In insulin signaling, these steps translate into insulin binding, receptor phosphorylation, IRS activation, kinase cascade activation, and metabolic enzyme modulation. Each step requires specific molecular interactions and proper timing for optimal pathway function.
What Happens During The Transduction Stage?
During transduction, the initial insulin-receptor binding event gets converted into intracellular phosphorylation cascades that amplify and distribute the signal throughout the cell. This stage involves the sequential activation of multiple protein kinases, each phosphorylating downstream targets to create branching signaling networks that ultimately coordinate diverse metabolic responses.
What Size Gauge Needle Is Best For Insulin Injections?
The optimal needle gauge for insulin injections typically ranges from 28 to 32 gauge, with 30-31 gauge needles being most commonly recommended. These thin needles minimize injection discomfort while ensuring reliable insulin delivery. Needle length should be 4-8mm for most patients, though individual factors like body composition may influence the best choice for specific situations.

