Does Zepbound Need to Be Refrigerated? Complete Guide 2025
Does Zepbound need to be refrigerated becomes a crucial question once you receive your prescription for this weight management medication. Zepbound requires refrigeration between 36°F and 46°F (2°C to 8°C) when stored long-term to maintain its effectiveness. However, you can keep unopened Zepbound pens at room temperature up to 77°F (25°C) for a maximum of 21 days. Once you start using a pen, it remains stable at room temperature for up to 21 days before requiring disposal. Never freeze Zepbound, as freezing permanently damages the medication and makes it unsafe to use. Understanding proper storage prevents medication waste and ensures each dose works effectively for your weight management goals. Room temperature storage offers flexibility for daily use and travel, while refrigeration extends shelf life for unopened pens.
Understanding Zepbound Storage Requirements
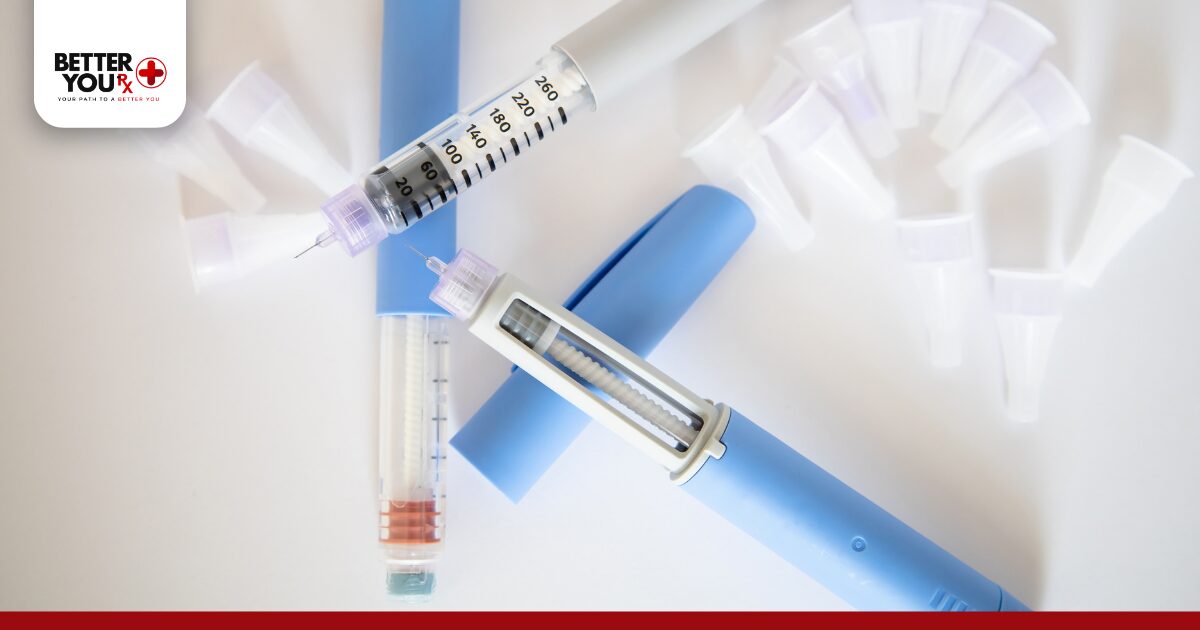
Zepbound contains tirzepatide, a medication that requires specific temperature conditions to remain effective. The active ingredient is a sensitive biological compound that breaks down when exposed to temperature extremes. Proper storage protects the medication’s potency and ensures safe, effective use.
Unopened Zepbound pens must stay refrigerated between 36°F and 46°F when stored for extended periods. This temperature range preserves the medication’s chemical structure and prevents degradation. Refrigeration extends the medication’s usable life until the expiration date printed on the pen.
The refrigerator’s main compartment provides ideal storage conditions. Avoid placing Zepbound in the freezer compartment or in areas that might freeze, such as near cooling vents. Keep the pens in their original carton to protect them from light exposure, which can also degrade the medication.
Store Zepbound away from the refrigerator door where temperature fluctuations occur more frequently. The constant opening and closing of the refrigerator door creates temperature variations that may affect medication stability. The middle or back shelves of the main compartment maintain more consistent temperatures.
Room temperature storage provides flexibility for active use. You can keep Zepbound at temperatures up to 77°F for 21 days maximum. This allowance makes daily dosing more convenient since you don’t need to handle cold medication during each injection.
Once you remove an unopened pen from the refrigerator for room temperature storage, you cannot return it to refrigerated conditions. The medication remains stable at room temperature for the 21-day window, but refrigerating it again after warming doesn’t reset this timeframe or extend usability.
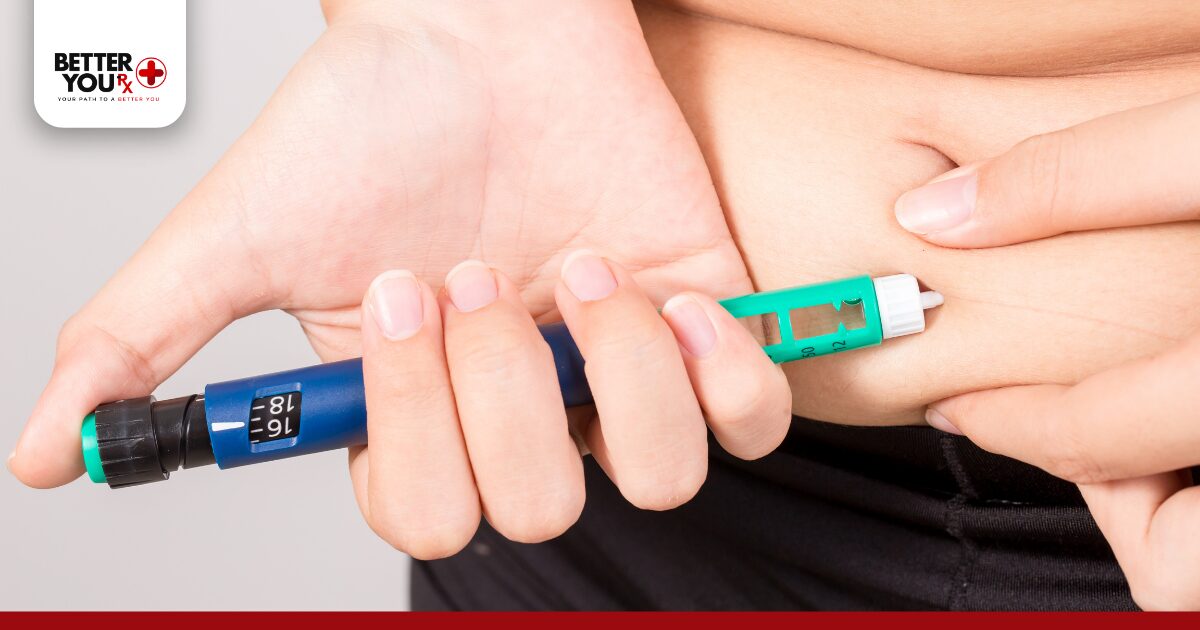
Does Zepbound Need to Be Refrigerated During Use
Once you start using a Zepbound pen, refrigeration becomes optional rather than required. You can store in-use pens either in the refrigerator or at room temperature up to 77°F, depending on your preference and convenience.
Many people find room temperature injections more comfortable than cold ones. Cold medication can cause temporary discomfort at the injection site, while room temperature Zepbound typically feels less noticeable during administration. Storing your active pen at room temperature eliminates the need to warm it before each injection.
The 21-day room temperature limit applies from the moment you first use the pen, not from when you remove it from the refrigerator. Track the date of your first injection and dispose of the pen 21 days later, even if medication remains inside. This timeframe ensures the medication maintains full potency throughout its use period.
Whether stored in the refrigerator or at room temperature, protect in-use pens from direct sunlight and heat sources. Avoid leaving Zepbound in cars, near windows, or in other locations where temperatures might exceed 77°F. Excessive heat accelerates medication breakdown and reduces effectiveness.
Keep the pen cap on when not in use to protect the medication from light and contamination. The cap creates a seal that maintains sterility and prevents accidental needle sticks. Replace the cap immediately after each injection.
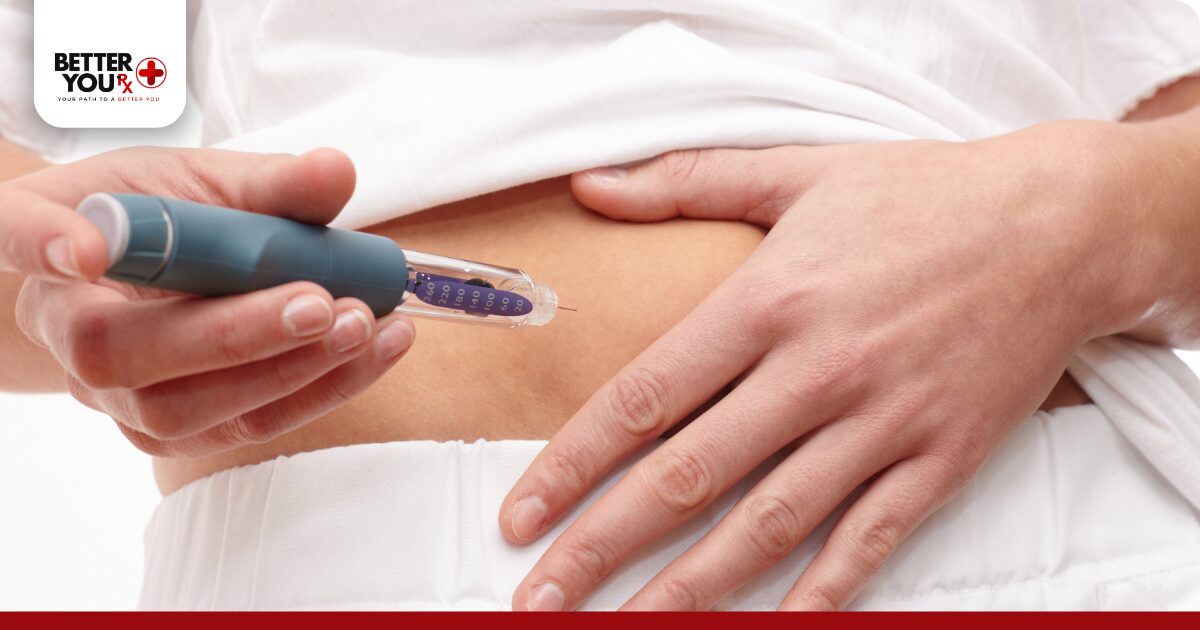
If you prefer refrigerated storage for in-use pens, allow the medication to reach room temperature before injecting. Remove the pen from the refrigerator 30 minutes before your scheduled dose. This warming period improves injection comfort and allows the medication to flow smoothly through the needle.
Ready to start your weight management journey? Order Zepbound KwikPen through Better You Rx, a prescription referral service offering convenient access to your medications.
ZepBound Room Temperature Storage Guidelines
Understanding room temperature storage limits helps you use Zepbound safely while maintaining flexibility in your daily routine. The 21-day room temperature window applies whether the pen is unopened or in use, giving you options for storage management.
Unopened Pen Storage
Unopened Zepbound pens can remain at room temperature up to 77°F for 21 consecutive days. This flexibility proves valuable when traveling or if refrigeration becomes temporarily unavailable. However, you must use or properly dispose of the pen within this timeframe.
Mark the date when you remove an unopened pen from the refrigerator. Write this date on the pen carton or keep a note in your phone to track the 21-day countdown. This practice prevents accidentally using expired medication that has exceeded room temperature storage limits.
Temperature consistency matters during room temperature storage. Avoid locations with significant temperature swings, such as bathrooms that get steamy or rooms that become very warm during the day. Stable temperatures between 68°F and 77°F provide optimal conditions.
In-Use Pen Storage
After your first injection from a pen, the 21-day room temperature clock starts regardless of where you store it. You can continue refrigerating the in-use pen or keep it at room temperature based on personal preference.
Calculate your disposal date from the first injection, not from when you started room temperature storage. For example, if you inject once weekly and take your first dose on January 1st, dispose of the pen on January 22nd, even if medication remains.
Some people find it helpful to label in-use pens with the first injection date and calculated disposal date. This clear marking prevents confusion about whether the pen remains usable, especially if you use multiple pens for different doses.
Temperature Monitoring
Consider using a room thermometer in the area where you store Zepbound at room temperature. This simple tool confirms that storage conditions stay within the acceptable range. Digital thermometers with min-max memory functions show whether temperatures exceeded safe limits while you were away.
During hot weather, indoor temperatures may exceed 77°F in homes without air conditioning. If your home gets too warm, store Zepbound in the coolest available location, use a cooler with ice packs for temporary storage, or return the medication to refrigeration if it hasn’t exceeded the 21-day room temperature limit.
Winter heating systems can also create temperature challenges. Avoid storing Zepbound near heating vents, radiators, or space heaters where localized temperatures might exceed safe limits even if the room feels comfortable overall.
Traveling With Zepbound
Travel requires extra planning to maintain proper storage conditions for Zepbound. Whether driving across town or flying internationally, you can transport your medication safely by following specific guidelines and using appropriate storage methods.
Short Trips and Daily Transport
For brief outings or daily activities, Zepbound remains stable without special storage equipment. If you’ll return home within a few hours, simply keep the medication in your bag away from direct heat sources. Avoid leaving Zepbound in hot cars, as vehicle interiors can quickly exceed safe storage temperatures.
When running errands on warm days, bring an insulated lunch bag if you’ll be away from home for several hours. Even without ice packs, the insulation provides some temperature protection. Place the medication in the center of the bag surrounded by other items for additional insulation.
Extended Travel and Vacation
Longer trips require more careful planning. Portable medication coolers designed for travel keep Zepbound within safe temperature ranges during extended journeys. These specialized coolers use ice packs, gel packs, or battery-powered cooling to maintain refrigeration temperatures.
Choose coolers specifically designed for medication storage rather than standard food coolers. Medication coolers provide more precise temperature control and often include temperature monitoring features. Look for models that maintain temperatures between 36°F and 46°F without freezing.
Pack ice packs around the Zepbound pen without allowing direct contact between the medication and frozen ice packs. Direct contact with ice can freeze the medication, permanently damaging it. Use a barrier like a small towel or the medication’s original packaging to prevent freezing.
Air Travel Considerations
Zepbound can travel in carry-on or checked luggage, though carry-on storage is generally preferable. Cabin temperatures remain more stable than cargo hold temperatures, and you maintain control over your medication throughout the journey.
Airport security allows medications in carry-on bags without size restrictions. Keep Zepbound in its original packaging with the prescription label visible. Inform security officers that you’re carrying refrigerated medication if they need to inspect your bag.
Consider the total travel time when planning air travel with Zepbound. If your journey including layovers exceeds a few hours, use a portable cooler to maintain appropriate temperatures. Many airport lounges provide refrigeration if you need to cool your medication during extended layovers.
Hotel and Accommodation Storage
Upon reaching your destination, immediately refrigerate Zepbound if you’ll be staying multiple days. Most hotel rooms include mini-fridges, but verify this amenity when booking. If no refrigerator is available, request one from the front desk or continue using your portable cooler with fresh ice packs.
Check the mini-fridge temperature if possible, as these units sometimes run warmer than standard refrigerators. If the refrigerator feels too warm or you’re uncertain about its temperature, continuing room temperature storage within the 21-day limit may be safer than using an inadequate refrigerator.
Planning a trip? Get your medications delivered before you travel from Better You Rx, a prescription referral service that makes medication access simple and convenient.
What Happens With Improper Storage
Understanding the consequences of improper storage helps you recognize when Zepbound may no longer be safe or effective. Temperature violations, freezing, and extended room temperature exposure all compromise medication quality.
Temperature Exposure Above 77°F
When Zepbound experiences temperatures above 77°F, the medication begins degrading more rapidly. Brief exposure to slightly elevated temperatures may not completely ruin the medication, but extended heat exposure significantly reduces potency.
If Zepbound sits in temperatures above 77°F for more than a few hours, the medication’s effectiveness becomes questionable. The tirzepatide compound breaks down under heat stress, potentially resulting in reduced weight management effects even if the medication appears normal.
Heat-damaged Zepbound shows no visible signs of degradation. The liquid doesn’t change color, develop particles, or smell different when heat-damaged. This lack of obvious indicators makes temperature monitoring crucial for ensuring medication quality.
Freezing Damage
Freezing permanently damages Zepbound and makes it completely unusable. The freezing process disrupts the medication’s molecular structure in ways that cannot be reversed by thawing. Frozen Zepbound must be discarded immediately, even if it appears normal after thawing.
Check for freezing if you suspect your Zepbound experienced temperatures below 32°F. Look for ice crystals inside the pen or on the medication’s surface. If the medication was completely frozen and has thawed, discard it regardless of appearance.
Partial freezing can be difficult to detect. If you’re uncertain whether Zepbound froze, examine the liquid carefully for any cloudiness or particles that weren’t present before. When in doubt, contact your healthcare provider or pharmacist for guidance rather than using potentially damaged medication.
Extended Room Temperature Storage
Zepbound left at room temperature beyond 21 days loses potency even if temperatures stayed within the acceptable range. The 21-day limit exists because the medication gradually degrades at room temperature, eventually becoming less effective.
Using Zepbound after the 21-day room temperature limit may not cause harm, but it likely won’t provide full therapeutic benefits. The reduced potency means you might not achieve expected weight management results despite continuing injections.
Track room temperature storage time carefully and dispose of pens that exceed the 21-day limit. Don’t try to extend usability by returning the medication to the refrigerator. Once the room temperature countdown begins, refrigeration doesn’t pause or reset the timeline.
Signs of Compromised Medication
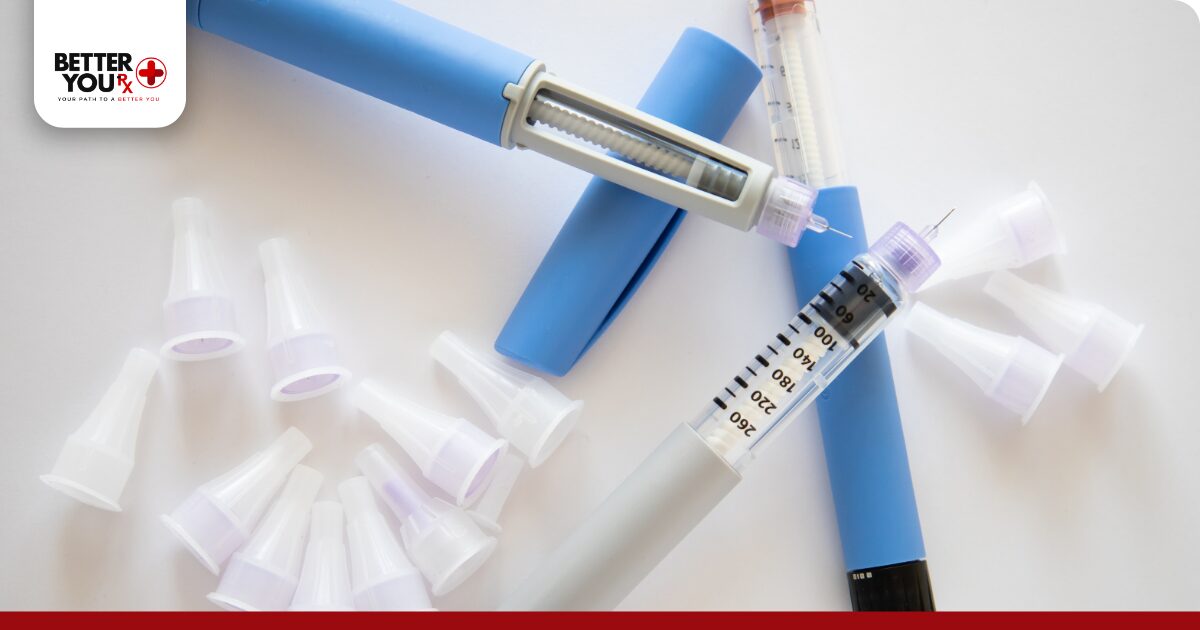
Inspect Zepbound before each use for signs of damage or contamination. Properly stored medication appears clear and colorless without particles, cloudiness, or discoloration. Any visible changes indicate the medication should not be used.
Discard Zepbound if you notice:
- Cloudiness or haziness in the liquid
- Particles, flakes, or floating debris
- Color changes from clear to yellow, brown, or any other tint
- Cracks or damage to the pen itself
- The medication has been frozen
- Room temperature storage exceeded 21 days
- You’re uncertain about storage conditions
When uncertain about whether Zepbound remains safe to use, err on the side of caution and request a replacement. Using compromised medication provides no benefits and may delay your treatment progress.
Best Practices for Zepbound Storage
Implementing consistent storage habits ensures your Zepbound remains effective throughout its usable life. These practical strategies minimize storage errors and help you maintain optimal medication quality.
Organizing Your Storage Space
Dedicate a specific location in your refrigerator for Zepbound storage. This consistent placement prevents accidentally misplacing the medication and makes it easy to grab when needed. The middle shelf typically provides the most stable temperature.
Store Zepbound in its original carton even while refrigerated. The carton protects against light exposure and provides space to write important dates like when you started room temperature storage or when an in-use pen expires.
Keep a small calendar or note near your storage location to track important dates. Record the date you started using each pen and calculate the disposal date 21 days later. This visible reminder prevents accidentally using expired medication.
Creating Storage Routines
Establish consistent habits around medication storage. Check the temperature of your refrigerator monthly using a refrigerator thermometer. This proactive monitoring catches temperature problems before they compromise your medication.
After each injection, immediately return the pen to its designated storage location. This habit prevents accidentally leaving the medication out where it might experience temperature extremes or get lost.
When starting a new pen, transfer any tracking information from the old pen to the new one. Note the first injection date on the pen carton and set a reminder for 21 days later. These redundant tracking methods prevent confusion about medication status.
Preparing for Emergencies
Keep backup cooling options available for power outages or refrigerator failures. A small cooler and reusable ice packs provide temporary storage during emergencies. Store ice packs in your freezer so they’re ready when needed.
During power outages, a refrigerator maintains safe temperatures for several hours if you keep the door closed. Avoid opening the refrigerator unnecessarily during outages to preserve cold air. If power will be out for extended periods, transfer Zepbound to a cooler with ice packs.
Have your healthcare provider’s contact information readily available for storage questions or emergencies. Quick access to professional guidance helps you make informed decisions when storage problems arise.
Managing Multiple Pens
If you have several Zepbound pens, organize them by expiration date with the oldest in front. This rotation system ensures you use pens before they expire and reduces waste from expired medication.
Label each pen clearly if you’re managing multiple doses or sharing refrigerator space with family members using the same medication. Clear labeling prevents mix-ups and ensures each person uses their own prescribed supply.
Consider the storage timeline when ordering refills. Order new supplies with enough advance time to receive and refrigerate them before your current supply expires, but not so far ahead that pens approach their expiration dates before use.
Need help managing your prescriptions? Visit Better You Rx, a prescription referral service dedicated to making medication access easier and more affordable.
ZepBound Storage Myths and Facts

Several misconceptions about Zepbound storage persist despite clear guidance. Understanding the facts helps you store your medication properly and avoid common mistakes that compromise effectiveness.
Myth – You Can Refreeze Thawed Zepbound
Some people believe that if Zepbound accidentally freezes, allowing it to thaw completely makes it usable again. This is false. Freezing causes irreversible damage to the medication’s molecular structure. Once Zepbound freezes, it must be discarded regardless of whether it appears normal after thawing.
The freeze-thaw process disrupts the delicate protein structure of tirzepatide. Even if the medication looks clear and normal after thawing, its effectiveness is permanently compromised. Never attempt to use Zepbound that has been frozen.
Myth – Refrigeration Resets the Room Temperature Clock
Another common misconception suggests that returning room temperature Zepbound to the refrigerator pauses or resets the 21-day countdown. This is incorrect. Once Zepbound begins room temperature storage, the 21-day clock continues regardless of whether you refrigerate it again.
The medication degradation that occurs during room temperature storage is cumulative. Refrigeration after warming doesn’t reverse degradation that already happened. Track total room temperature exposure time, not just current storage conditions.
Myth – Cold Medication Works Better
Some users believe that injecting cold Zepbound directly from the refrigerator makes it more effective. Temperature at injection doesn’t affect medication potency. Cold injections simply feel less comfortable than room temperature ones.
The medication’s effectiveness depends on proper storage throughout its life, not its temperature at the moment of injection. Allowing refrigerated Zepbound to warm to room temperature before injecting improves comfort without affecting results.
Fact – Sunlight Damages Zepbound
Direct sunlight exposure can degrade Zepbound even if the temperature remains within acceptable ranges. Light-induced degradation occurs separately from temperature damage. Always store Zepbound in its original carton or another opaque container to protect it from light.
Window sills, countertops near windows, and other sun-exposed areas are unsuitable storage locations even if they feel cool. The combination of light and moderate warmth accelerates medication breakdown.
Fact – Storage Affects Your Results
Proper storage directly impacts your weight management outcomes. Degraded medication delivers reduced amounts of active tirzepatide, potentially resulting in diminished appetite suppression and weight management effects. Consistent proper storage ensures you receive full therapeutic benefits from each dose.
If you notice reduced effectiveness from your Zepbound injections, review your storage practices. Improper storage may explain unexpected changes in medication response.
Key Takeaways
Zepbound requires refrigeration between 36°F and 46°F for long-term storage, but offers flexibility with 21 days of room temperature storage up to 77°F. This combination of requirements and allowances makes Zepbound manageable for daily use while ensuring medication stability.
Never freeze Zepbound, as freezing causes permanent damage that makes the medication unusable. Store pens in the main refrigerator compartment away from freezing zones and temperature-variable areas like the door. Keep Zepbound in its original carton to protect against light exposure.
Track room temperature storage time carefully, starting the 21-day countdown from when you first remove the pen from refrigeration or take your first injection, whichever comes first. Dispose of pens after 21 days at room temperature, even if medication remains inside.
Travel with Zepbound using insulated bags or portable medication coolers for trips lasting several hours. Maintain temperature awareness during travel and immediately refrigerate the medication upon reaching your destination if staying multiple days.
Inspect Zepbound before each use for signs of damage like cloudiness, particles, or discoloration. Discard any medication that appears abnormal or if you’re uncertain about its storage history. Proper storage practices ensure each dose delivers full effectiveness for your weight management goals.
Frequently Asked Questions
What Happens if I Don’t Refrigerate Zepbound?
Unopened Zepbound can remain at room temperature up to 77°F for 21 days without refrigeration. After 21 days at room temperature, the medication begins losing potency and should be discarded. If you accidentally left unopened Zepbound unrefrigerated, check how long it was at room temperature. If less than 21 days and temperatures stayed below 77°F, the medication remains usable. Mark the date you discovered it was unrefrigerated and calculate 21 days from when it likely left refrigeration. If you’re uncertain how long it was unrefrigerated or if it exceeded 21 days, request a replacement rather than risk using compromised medication.
How to Keep Zepbound Cold While Traveling?
Keep Zepbound cold while traveling using insulated medication coolers with ice packs or gel packs. Choose coolers designed specifically for medication that maintain temperatures between 36°F and 46°F. Place ice packs around the Zepbound pen without direct contact to prevent freezing. For air travel, pack Zepbound in carry-on luggage where you control storage conditions. Most flights provide temperatures suitable for the 21-day room temperature allowance, but longer international trips benefit from portable cooling. When driving, never leave Zepbound in a parked car where temperatures can quickly exceed safe limits. Bring a small cooler even for day trips during hot weather.
What Happens to Zepbound After 21 Days?
After 21 days at room temperature, Zepbound begins degrading and losing effectiveness. The active ingredient tirzepatide breaks down gradually at room temperature, and the 21-day limit represents the point where potency loss becomes significant. Using Zepbound after 21 days at room temperature likely won’t cause harm, but it may not provide expected weight management benefits. The reduced potency means you might not experience full appetite suppression or weight loss effects despite continuing injections. Always dispose of Zepbound that exceeded the 21-day room temperature limit and start with a fresh pen to ensure optimal results.
What Happens if Tirzepatide Is Not Refrigerated?
Tirzepatide, the active ingredient in Zepbound, remains stable at room temperature up to 77°F for 21 days. Beyond this timeframe or at higher temperatures, tirzepatide begins breaking down and losing effectiveness. The medication doesn’t become harmful when left unrefrigerated within reasonable limits, but its potency decreases. Short-term lack of refrigeration within the 21-day window doesn’t damage tirzepatide if temperatures stayed below 77°F. However, extended unrefrigerated storage or exposure to high temperatures compromises the medication’s ability to provide weight management benefits. When uncertain about storage conditions, replace the medication rather than risk using ineffective tirzepatide.
Can I Carry Zepbound on a Plane?
You can carry Zepbound on a plane in either carry-on or checked luggage, though carry-on storage is preferable. Carry-on storage allows you to monitor medication conditions throughout your flight and prevents exposure to extreme cargo hold temperatures. Airport security permits medications in carry-on bags regardless of liquid volume restrictions. Keep Zepbound in its original packaging with the prescription label visible for security screening. Inform security officers you’re carrying refrigerated medication if needed. For flights lasting several hours, consider using a small insulated bag with ice packs to maintain appropriate temperatures, though the 21-day room temperature allowance covers most travel scenarios without special cooling equipment.
Is Tirzepatide Still Good if Left Out Overnight?
Tirzepatide remains good if left out overnight as long as room temperature stayed below 77°F and the total room temperature storage time doesn’t exceed 21 days. One night at room temperature doesn’t damage the medication. However, start tracking the 21-day countdown from when you left it out if this was an unopened pen’s first room temperature exposure. If the pen was already in use or previously at room temperature, add the overnight hours to your cumulative room temperature time. Check that the medication appears clear and colorless before your next injection. If anything looks abnormal or you’re concerned about storage conditions, contact your healthcare provider for guidance.
Does Zepbound Need to Be Refrigerated After Opening?
Zepbound does not require refrigeration after opening, though you can continue refrigerating in-use pens if preferred. Once you take your first injection from a pen, you can store it at room temperature up to 77°F for 21 days total. Many people find room temperature storage more convenient for active pens since it eliminates the need to warm the medication before each injection. Whether you refrigerate or use room temperature storage, the 21-day disposal timeline starts from your first injection. After 21 days, discard the pen even if medication remains inside. Choose whichever storage method feels most convenient while staying within the 21-day limit and temperature requirements.




 Does Wegovy make you tired is a common concern among people starting this weight loss medication. Fatigue represents one of the frequently reported experiences during Wegovy treatment, though it varies significantly among users. The tiredness typically stems from reduced calorie intake, changes in metabolism, dehydration, nutritional adjustments, and your body adapting to the medication. Most people experience fatigue during the first few weeks of treatment as their system adjusts to appetite changes and lower food consumption. The good news is that this tiredness usually improves over time as your body adapts. Managing fatigue involves staying hydrated, eating nutrient-dense foods, maintaining gentle exercise, getting adequate sleep, and adjusting meal timing.
Does Wegovy make you tired is a common concern among people starting this weight loss medication. Fatigue represents one of the frequently reported experiences during Wegovy treatment, though it varies significantly among users. The tiredness typically stems from reduced calorie intake, changes in metabolism, dehydration, nutritional adjustments, and your body adapting to the medication. Most people experience fatigue during the first few weeks of treatment as their system adjusts to appetite changes and lower food consumption. The good news is that this tiredness usually improves over time as your body adapts. Managing fatigue involves staying hydrated, eating nutrient-dense foods, maintaining gentle exercise, getting adequate sleep, and adjusting meal timing.