Why Increase Zepbound Dose? Understanding the Step-Up Plan
Zepbound (Tirzepatide) follows a structured dose escalation approach that gradually increases medication strength over time. This method allows your body to adapt while minimizing potential side effects during treatment. The step-up plan typically begins at 2.5 mg weekly and progresses through higher doses based on your response and weight management goals. Each dose level serves a specific purpose in your treatment journey, with increases scheduled at four-week intervals. The gradual progression helps maintain effectiveness while supporting sustainable weight loss results. Understanding when and why doses increase helps you prepare for each phase of treatment. Most patients move through multiple dose levels before reaching their optimal maintenance dose, which varies based on individual response and health objectives. This step-by-step approach ensures your body adjusts properly while delivering consistent results throughout your weight loss journey.
How Your Body Adjusts to Higher Zepbound Doses

Gradual dose increases work because your body needs time to get used to how Tirzepatide affects your system. Starting with smaller amounts and slowly increasing helps you avoid uncomfortable reactions while still getting good results.
Initial Body Response
When you first start taking Zepbound, your digestive system begins responding to the medication’s effects. The drug slows down how quickly food moves through your stomach and changes the signals your brain receives about hunger and fullness. These changes don’t happen all at once, which is why starting at a lower dose makes sense.
The beginning 2.5 mg dose helps your body learn how to work with the medication. This starting amount isn’t meant to be your final dose – it’s more like a gentle introduction that gets your metabolism ready for the treatment ahead. During these first weeks, your body starts activating the processes needed for weight loss without overwhelming your system. Your digestive system, hormone levels, and appetite signals all begin shifting in response to the medication. This early phase establishes the foundation for everything that comes next in your treatment.
Building Tolerance Over Time
After about four weeks, your body becomes familiar with the current dose. This adjustment means you’re ready to move up to the next level without experiencing strong side effects. The monthly timing between increases gives enough time to see how each dose works for you before adding more medication. Your body has fully adapted to the current strength, and staying at that level would likely result in diminishing returns as your system becomes too comfortable with the medication.
Studies show that people who increase doses slowly have better success over time compared to those who try to start at higher amounts right away. Patients following the step-up plan are less likely to stop treatment early and tend to lose weight more consistently throughout their journey. The gradual approach reduces the shock to your system and allows your body to build tolerance in a controlled, manageable way. This means fewer disruptions to your daily life and better adherence to your treatment schedule.
Progressive Adaptation Process
Your body builds tolerance at each level, which is a normal part of how medication works. This tolerance doesn’t mean the treatment stops working – it means you’re ready for the next step that will continue delivering results. The planned increases stay ahead of this adjustment process, keeping your weight loss moving forward. Think of tolerance as your body’s way of signaling that it can handle more now. What once felt like a strong effect becomes your new normal, indicating readiness for advancement.
Each dose level prepares your system for the next one. Think of it like building strength at the gym – you don’t start by lifting the heaviest weights. You begin with manageable amounts and gradually increase as your body becomes stronger. Zepbound dosing follows this same sensible approach. Your digestive system becomes more efficient at processing the medication, your hormone receptors become more responsive, and your body learns to maintain the metabolic changes even as doses increase.
Developing Healthy Habits
The step-up method also gives you time to develop new eating habits at each stage. As your appetite decreases with each dose increase, you naturally learn to eat smaller portions and make better food choices. These behavioral changes become ingrained over time, setting you up for success even after you complete your treatment. The medication creates the window of opportunity, but the habits you build during the dose escalation process determine your long-term results.
Standard Zepbound Dosing Schedule
The medication follows a predetermined escalation path designed for optimal results. Each patient begins at the same starting point regardless of their weight loss goals or medical history. This standardized approach ensures safety while allowing for individual variations in how quickly people progress through the levels.
Week 1-4 – Initial Phase
Starting dose at 2.5 mg weekly marks the beginning of your treatment journey. The primary focus during this month centers on body adaptation and tolerance assessment. You’ll likely notice mild appetite reduction and initial metabolic changes as your body responds to the medication. This phase involves gastrointestinal adjustment as your digestive system learns to work with Tirzepatide. Most people experience some stomach sensitivity during these first weeks, which is completely normal and expected. The key is giving your body the full four weeks to adjust before moving forward.
Week 5-8 – First Increase
Dose level increases to 5 mg weekly as you enter the second month of treatment. Treatment goals shift toward enhanced appetite control and steady weight reduction. Physical changes become more noticeable, with stronger fullness signals and natural portion size reduction happening almost automatically. The adjustment period continues as your GI system develops improved tolerance to the medication. Many patients report that this is when weight loss becomes more consistent and predictable. Your body has moved past the initial shock and now works efficiently with the medication.
Week 9-12 – Second Increase
Dose level moves up to 7.5 mg weekly during your third month. Therapeutic effects accelerate with improved metabolic response and sustained weight loss becoming evident. Body response shows established eating pattern changes and reduced cravings for high-calorie foods. Maintenance considerations begin as you and your provider assess whether this level provides adequate results or if further increases would benefit your progress. Some patients find their sweet spot at this dose and may stay here longer before advancing. Others notice their body is ready to keep climbing the dose ladder.
Week 13-16 – Third Increase
Dose level reaches 10 mg weekly as you complete your fourth month of treatment. Enhanced outcomes appear through maximized receptor activation and metabolic optimization throughout your body. Physical indicators include consistent weight loss trends and well-regulated appetite that feels natural rather than forced. Progress evaluation becomes important during this phase to review treatment effectiveness and tolerance levels. Your provider will likely want detailed feedback about how you’re feeling, what results you’re seeing, and whether side effects remain manageable. This checkpoint helps determine the path forward.
Week 17 and Beyond – Higher Dose Options
Available levels of 12.5 mg and 15 mg weekly provide options for sustained weight management and plateau prevention. The purpose of these higher doses focuses on helping patients who need additional support to reach their goals. Selection criteria depend on individual response patterns and remaining weight loss targets that haven’t been achieved at lower levels. Long-term strategy involves determining your maintenance dose for continued success after the active weight loss phase ends. Not everyone needs these maximum doses, but they’re available for those whose bodies require them to achieve optimal results.
Looking to start your weight management journey? Better You Rx connects you with licensed healthcare providers who can evaluate your needs and create a personalized treatment plan.
Why Increase Zepbound Dose Over Time
The progressive increase strategy addresses multiple physical and treatment factors that emerge during your weight loss journey. Your body’s response to medication changes as treatment continues, requiring dose adjustments to maintain effectiveness.
Metabolic Adaptation
Your metabolism naturally adapts as you continue taking the same dose week after week. The parts of your body that respond to Tirzepatide become somewhat used to the medication, which means the initial dose doesn’t work as strongly over time. Increasing the dose keeps these systems active and working properly for continued weight loss. This adaptation happens in your digestive tract, your brain’s hunger centers, and your body’s fat-burning processes. Without dose increases, these adaptations would eventually slow or stop your progress entirely.
Overcoming Weight Loss Plateaus
Weight loss plateaus represent another key reason for dose increases. As you lose weight, your body’s calorie-burning rate changes to match your new size. The dose that helped you lose weight initially may not produce the same results as your body composition changes. Higher doses give you back that advantage so you can keep making progress toward your goals. Your smaller body requires fewer calories to maintain itself, so the appetite suppression needs to be stronger to continue creating the calorie deficit necessary for ongoing weight loss.
Enhanced Appetite Control
Controlling your appetite requires ongoing medication support at the right strength. The starting dose provides basic appetite control, but losing more weight needs stronger signals to help you eat less. Each dose increase strengthens the fullness signals and helps you feel satisfied longer between meals. This enhanced satiety makes it easier to stick with smaller portions and resist cravings that might otherwise derail your progress. The medication does the heavy lifting while you focus on building healthy habits.
Individual Response Variations
Everyone responds to medication differently, which is why flexible dosing makes sense. Some people get great results at lower doses, while others need the maximum strength to reach their weight loss targets. The step-up plan works for these differences by offering several dose options throughout your treatment. Your genetics, metabolism, starting weight, and lifestyle factors all influence how you respond to each dose level. The structured increases ensure everyone has the opportunity to find their optimal dose.
Timing and Momentum
The timing of dose increases matters for keeping your progress steady. Waiting too long at a lower dose when your body is ready for more can slow down your results. Moving up at the right time keeps your weight loss momentum going without unnecessary delays. Conversely, rushing through doses before your body has fully adapted can trigger uncomfortable side effects that might force you to pause or reduce your dose. The four-week intervals represent the sweet spot for most patients.
Behavioral Changes Support
Your eating habits change as doses increase. Higher amounts help you naturally eat smaller portions and feel full faster. These changes support the behavioral adjustments needed for long-term weight management success beyond just taking medication. You learn to recognize true hunger versus boredom or emotional eating. You discover that smaller amounts of food actually satisfy you when you eat slowly and mindfully. These lessons stay with you even after treatment ends.
Fat Metabolism Enhancement
The medication also impacts how your body stores and burns fat. Higher doses activate more fat-burning processes while reducing fat storage signals. This metabolic shift becomes more pronounced as doses increase, contributing to accelerated weight loss in the later stages of treatment. Your body becomes more efficient at using stored fat for energy, which is exactly what you want when trying to lose weight.
| Dose Level | Primary Purpose | Typical Duration | Expected Outcomes |
| 2.5 mg | Treatment start and tolerance building | 4 weeks | Initial appetite changes, 2-3% weight reduction |
| 5 mg | Active treatment and steady progress | 4 weeks | Better appetite control, 4-6% total weight loss |
| 7.5 mg | Metabolism boost and continued momentum | 4 weeks | Stronger fullness signals, 7-10% total weight loss |
| 10 mg | Sustained effectiveness and avoiding plateaus | 4+ weeks | Consistent progress, 10-14% total weight loss |
| 12.5 mg | Advanced weight management support | Ongoing | Maintained momentum, 14-18% total weight loss |
| 15 mg | Maximum strength benefit | Maintenance | Peak effectiveness, 18-22%+ total weight loss |
Signs You’re Ready for the Next Dose Level

Recognizing readiness for dose increases involves watching for specific signals that show your body has adjusted to the current level. These signs help confirm that moving up will benefit your treatment progress rather than causing unnecessary problems.
Digestive System Indicators
Your digestive system gives clear signals when adjustment happens. Starting side effects like mild nausea or bathroom changes typically get better after two to three weeks at each dose level. When these effects go away completely, your body shows it’s ready for advancement. You’ll notice that foods you avoided during the initial adjustment period no longer bother you. Your stomach feels settled and comfortable most of the time. This stability indicates your GI system has fully adapted to the current medication strength.
Appetite Pattern Changes
Appetite patterns shift as tolerance develops at each dose. You may notice hunger returning closer to how it was before treatment, or feeling less satisfied after meals than you did during the first weeks at this dose. These changes indicate that the current dose no longer provides strong enough appetite suppression, suggesting potential benefit from a higher concentration. You might find yourself thinking about food more often or needing larger portions to feel full. These are natural signs of adaptation, not treatment failure.
Weight Loss Progress Markers
Weight loss progression provides important markers for readiness. Consistent weight loss at the current dose level indicates proper medication response. When you experience steady reduction for three to four weeks, this establishes a baseline for comparison. A subsequent slowdown or plateau in weight loss, despite maintained lifestyle habits, often signals the need for dose adjustment. If you’re doing everything right with diet and exercise but the scale stops moving, your body is likely ready for more medication support.
Energy and Physical Capacity
Energy levels and physical capacity improve as treatment progresses. Patients ready for dose increases typically report stable energy throughout the day and improved exercise tolerance. These positive changes confirm that your body handles the current dose well without adverse effects limiting daily activities. You feel strong enough to increase your physical activity, which is a great sign that the medication is working with your body rather than against it.
Timeline Considerations
The standard four-week interval between doses exists for specific reasons. Advancing before this timeframe prevents accurate assessment of each dose level’s full effects. Your body needs the complete month to show its true response pattern. Some people feel ready to increase sooner, but patience during this period pays off with smoother transitions and better results. Waiting beyond four weeks without good medical reasons may delay optimal results and extend overall treatment duration unnecessarily.
Mental and Emotional Readiness
Your mental state also reflects readiness for dose advancement. Feeling confident and positive about your progress indicates you’re handling the current dose well psychologically. If you’re anxious, overwhelmed, or struggling with the lifestyle changes, you might benefit from staying at the current level a bit longer to build more confidence before advancing. The medication works best when your mental and physical readiness align.
Sleep Quality Improvements
Sleep quality often improves as your body adjusts to each dose. When you’re sleeping well consistently, waking up refreshed, and maintaining good energy during the day, these signs suggest your body has adapted fully to the current strength. Poor sleep or fatigue that persists beyond the first two weeks might indicate you need more time at your current dose before moving up.
Ready to take the next step in your treatment? Order Zepbound Kwikpen through Better You Rx, a prescription referral service offering access to quality medications at competitive rates.
Managing Side Effects During Dose Increases
Each dose escalation brings the possibility of renewed side effects as your body adjusts to higher medication concentrations. Understanding how to manage these effects supports successful progression through the treatment plan without unnecessary discomfort or disruption to your daily life.
Digestive Changes and Nausea
Digestive changes represent the most common side effects during dose increases. Nausea typically appears within the first few days after advancing to a new level and gradually subsides over one to two weeks. This pattern repeats with each increase, though many people find that later increases cause less nausea than earlier ones as their body becomes more accustomed to the medication’s effects. The nausea usually feels worst in the morning or after eating, and tends to improve as the day goes on.
Eating Strategies for Comfort
Eating smaller, more frequent meals throughout the day helps manage nausea and digestive discomfort. Instead of three large meals, try five or six small eating occasions spread across your waking hours. This approach keeps your stomach from getting too empty or too full, both of which can trigger nausea.
- Choose bland, easily digestible foods during adjustment periods – think plain rice, toast, bananas, applesauce, and lean proteins prepared simply
- Avoid high-fat or heavily spiced dishes that may trigger discomfort
- Stay upright for at least 30 minutes after eating to help your stomach process food more comfortably
- Prop yourself up with pillows if you need to rest rather than lying flat
Hydration Management
Hydrate consistently with small sips of water or clear fluids throughout the day. Dehydration worsens nausea and other side effects, but drinking large amounts at once can make you feel uncomfortably full. Keep a water bottle nearby and take small sips regularly. Some people find that slightly cool water or adding a slice of lemon helps settle their stomach. Avoid sugary drinks, caffeine, and alcohol during the adjustment period as these can irritate your digestive system.
Bowel Habit Adjustments
Bowel habit changes may occur as your digestive system responds to increased medication levels. Some patients experience looser stools while others notice constipation during the adjustment phase.
- For loose stools, stick with low-fiber foods temporarily and avoid dairy if it bothers you
- For constipation, increase water intake, add gentle fiber sources like oats and vegetables, and stay physically active
- Most bowel changes resolve within two weeks as your body adjusts
Nutrition Despite Reduced Appetite
Higher doses intensify appetite suppression, sometimes making adequate nutrition challenging. Focus on nutrient-dense foods that provide essential vitamins and minerals in smaller volumes. Even though you’re not hungry, your body still needs proper nutrition to function well and maintain muscle mass during weight loss. Protein-rich options support muscle maintenance and promote satiety, so prioritize foods like Greek yogurt, eggs, chicken, fish, and legumes. Don’t skip meals just because you’re not hungry – your body needs consistent fuel.
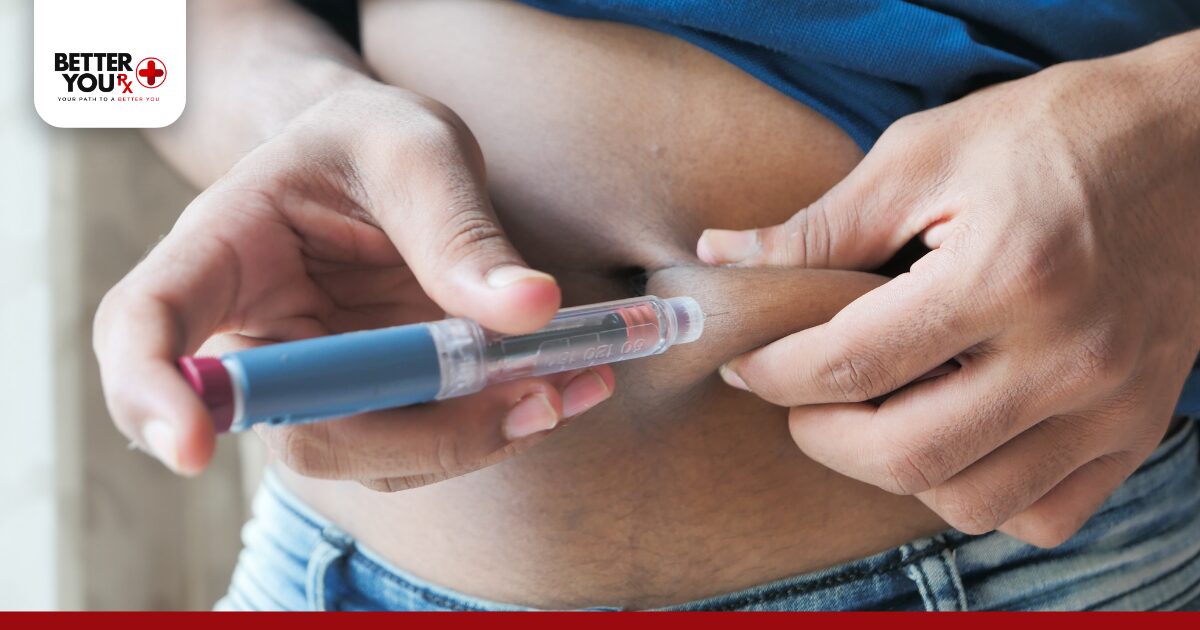
Injection Timing Optimization
Timing your injection can influence side effect intensity. Many patients find that administering their dose in the evening reduces daytime nausea and digestive discomfort. The strongest side effects typically occur within 24-48 hours after injection, so evening dosing means these effects happen mostly while you’re sleeping or less active. Experiment with injection timing to identify the schedule that minimizes interference with work, social activities, and daily responsibilities. Some people prefer morning injections to maintain a consistent routine, while others swear by evening administration for better tolerance.
When to Seek Medical Help
Certain symptoms require professional evaluation rather than home management. Persistent vomiting that prevents adequate hydration represents a serious concern that needs immediate medical attention. Severe abdominal pain that doesn’t respond to basic comfort measures or that worsens over time should be evaluated. Symptoms that continue getting worse instead of improving after two weeks at a new dose warrant discussion with your provider, as this pattern suggests possible intolerance rather than normal adjustment.
Recognizing Unusual Symptoms
Unusual symptoms not typically associated with Tirzepatide also merit discussion with your provider. These include persistent headaches that don’t respond to over-the-counter pain relievers, vision changes like blurriness or seeing spots, or signs of allergic reaction like rash, itching, or breathing difficulties. While these symptoms are rare, recognizing them early and getting proper evaluation ensures your safety throughout treatment.
Tracking and Pattern Recognition
Keep a side effect journal during dose increases to track patterns and identify what helps you feel better. Note the timing of symptoms, what you ate before they occurred, and which strategies provided relief. This information helps you predict and prevent problems while also giving your provider valuable data about your response to treatment. Many people find that their side effects follow predictable patterns, allowing them to plan around them.
Factors That Influence Dose Progression Speed

Not every patient follows the standard escalation timeline exactly. Several factors determine whether you progress through doses at the typical pace or require modifications to the schedule based on your unique circumstances and response patterns.
Individual Tolerance Variations
Individual tolerance varies significantly among patients. Some people experience minimal side effects and tolerate each increase without difficulty, breezing through the dose escalation with barely any discomfort. Others require extended time at lower doses before their bodies adapt sufficiently for advancement. Neither pattern indicates treatment failure or superior response – they simply reflect natural variation in how different bodies process medication. Your tolerance level doesn’t predict your final results, so don’t worry if you need more time at each level than other patients seem to need.
Weight Loss Velocity Impact
Weight loss velocity influences dose progression decisions. Patients achieving excellent results at lower doses may remain at those levels longer before increasing. If you’re losing weight consistently and feeling good at your current dose, there’s no urgent need to advance just because the calendar says it’s been four weeks. Conversely, those experiencing slower progress might benefit from advancing to higher doses more quickly to enhance effectiveness. Your provider will help balance these considerations based on your individual goals and response.
Medical History Considerations
Medical history and concurrent health conditions affect dosing strategies. People with certain digestive conditions may need gradual increases with extended adaptation periods between levels. Those with thyroid issues, diabetes, or other metabolic conditions might progress through lower doses more rapidly or slowly based on their unique body chemistry. Previous experience with weight loss medications can also influence how your provider approaches dose escalation. Always share your complete medical history so your provider can tailor the approach to your specific needs.
Lifestyle Factor Influence
Lifestyle factors including diet quality, physical activity levels, and stress management impact how well you tolerate dose increases. Patients who maintain consistent healthy habits typically experience smoother transitions between dose levels with fewer side effects.
- Regular exercise helps your body process the medication more efficiently and reduces nausea for many people
- Good sleep hygiene supports your body’s adjustment processes
- High stress levels can amplify side effects and slow your progress, making stress management an important part of your treatment strategy
Treatment Goal Alignment
Treatment goals play a role in determining optimal dose progression. Someone with modest weight loss objectives might achieve their target at intermediate doses and choose to stop there rather than continuing to maximum strength. Another person with more ambitious goals may require maximum doses to reach their desired outcomes. Your personal finish line influences how aggressively you need to pursue dose increases. Discuss your specific targets with your provider so they can help map out the most efficient path.
Age and Metabolism Effects
Age and baseline metabolism affect how quickly your body adapts to each dose level. Younger patients with faster metabolisms sometimes need to progress more quickly to maintain effectiveness, while older patients might benefit from a more gradual approach. Neither is better or worse – it’s simply about matching the progression speed to your body’s needs and capabilities.
Weight Loss History
Previous weight loss attempts and yo-yo dieting history can influence dose requirements. Bodies that have been through multiple cycles of weight loss and regain sometimes require higher doses or slower progression to overcome metabolic adaptation. This doesn’t mean you can’t succeed – it just means your path might look different from someone attempting weight loss for the first time.
Support System Strength
Support system strength impacts your ability to manage side effects and stay consistent with treatment. Patients with strong family support, understanding employers, and helpful healthcare teams often tolerate dose increases better because they have help managing the adjustment period. If you’re navigating treatment mostly alone, you might need to progress more slowly to ensure each transition remains manageable within your daily life constraints.
Maximizing Results at Each Dose Level
Success with Zepbound extends beyond simply taking the medication as prescribed. Implementing supportive strategies at every dose level enhances effectiveness and promotes sustainable weight management that lasts well beyond your active treatment period.
Nutritional Optimization
Focus on whole, minimally processed foods that provide maximum nutritional value. Vegetables, lean proteins, whole grains, and healthy fats support your body during treatment while promoting steady weight loss. Plan meals in advance to avoid impulsive food choices when appetite suppression makes eating less appealing. When you’re not feeling hungry, it’s easy to skip meals or grab whatever’s convenient, but thoughtful planning ensures you get the nutrition your body needs. Prep healthy options over the weekend so they’re ready when you need them during busy weekdays.
Protein Prioritization
Protein intake deserves special attention throughout treatment. Adequate protein preserves muscle mass during weight loss and supports metabolic function. Include protein sources at each meal, even when portions seem small compared to pre-treatment eating patterns. Aim for at least 60-80 grams of protein daily, distributed across your eating occasions. This might mean a Greek yogurt at breakfast, chicken salad at lunch, and fish with vegetables at dinner, plus protein-rich snacks like hard-boiled eggs or nuts.
Physical Activity Integration
Regular movement complements medication effects by boosting metabolism and supporting body composition changes. Start with activities you enjoy and can maintain consistently rather than intense workouts that prove unsustainable.
- Walking, swimming, cycling, dancing, or any activity that gets you moving counts toward your goals
- Consistency matters more than intensity, especially in the beginning
- Twenty minutes of daily walking beats an hour-long gym session you only manage once a week
Strength Training Benefits
Strength training becomes increasingly important as treatment progresses. Resistance exercises maintain muscle tissue that might otherwise decrease during calorie restriction. Even modest strength work twice weekly provides significant benefits for long-term weight maintenance. You don’t need a gym membership or fancy equipment – bodyweight exercises like squats, push-ups, and planks build strength effectively. Resistance bands offer another affordable option for home workouts.
Hydration Priorities
Water intake supports medication effectiveness and helps manage potential side effects. Aim for adequate fluid consumption throughout the day, adjusting based on activity level and climate. Proper hydration also supports digestive function during dose increases and helps your body process the medication efficiently. Many people find that staying well-hydrated reduces nausea and other side effects. Keep water accessible throughout the day to make consistent hydration easier.
Sleep Quality Focus
Quality sleep influences weight loss hormones and appetite regulation in powerful ways. Prioritize consistent sleep schedules and create conditions that promote restful nights. The metabolic benefits of adequate sleep enhance Tirzepatide’s effectiveness at every dose level. Poor sleep increases hunger hormones and reduces fullness signals, working against your medication. Aim for seven to nine hours nightly and maintain a regular sleep-wake schedule, even on weekends.
Mindful Eating Practices
Use the appetite reduction provided by medication to establish healthier eating patterns. Practice mindful eating, paying attention to true hunger signals versus emotional or habitual eating triggers. The medication window offers valuable time to build habits that support long-term success beyond active treatment.
- Learn to eat when you’re actually hungry rather than by the clock
- Recognize the difference between head hunger (driven by boredom, stress, or habit) and body hunger (actual physical need for food)
- Eat slowly and without distractions to fully experience your meals
Progress Monitoring Beyond the Scale
Monitor progress through multiple measures beyond scale weight alone. Take body measurements of your waist, hips, arms, and thighs monthly. Notice how clothing fits, paying attention to items that become looser even when the scale isn’t moving. Track non-scale victories like improved energy, better sleep, easier physical activities, or reduced medication needs for other conditions. These markers provide motivation during periods when weight loss temporarily plateaus.
Building Support Networks
Build a support network that understands your goals and encourages your efforts. Connect with others on similar journeys through online communities or local support groups. Share your challenges and victories with trusted friends or family members. Consider working with a registered dietitian or health coach who can provide personalized guidance. Having people who understand what you’re experiencing makes the journey feel less isolating and provides accountability.
Addressing Emotional Eating
Address emotional eating patterns during treatment while appetite suppression creates space for this work. Many people use food to cope with stress, boredom, sadness, or anxiety. The medication reduces the urgency of these eating urges, giving you the opportunity to develop healthier coping mechanisms. Explore alternatives like walking, journaling, calling a friend, or practicing relaxation techniques when emotional triggers arise.
Environment Optimization
Create an environment that supports your goals by removing temptations and stocking healthy options. Keep nutritious snacks visible and accessible while storing less healthy choices out of sight or eliminating them entirely. Prepare your space for success by having proper food storage containers for meal prep, a water bottle you enjoy using, and comfortable shoes for walking. Small environmental changes remove barriers to healthy choices.
Find affordable access to your prescribed medications at Better You Rx, where you can connect with quality treatment options.
The Bottom Line
The step-up dosing approach with Zepbound creates a foundation for sustainable weight management success. Each dose increase represents progress toward your health goals while allowing your body time to adapt throughout the journey. Understanding the reasons behind gradual escalation helps you approach each phase with realistic expectations and confidence in the process. The structure exists to protect your wellbeing while maximizing your results, not to frustrate you with slow progress.
Your individual path through dose levels may look different from others based on your unique response patterns and objectives. Some patients reach their goals at intermediate doses while others benefit from maximum strength to achieve desired outcomes. Both scenarios represent successful treatment when aligned with your specific needs and health targets. Don’t compare your journey to someone else’s – focus on your own progress and trust the process designed specifically to help you succeed.
Active participation in your treatment enhances results at every dose level. Combining medication with nutritious eating patterns, regular physical activity, and healthy lifestyle habits creates synergy that amplifies Tirzepatide’s effects. The medication provides powerful support, but your daily choices determine long-term success and maintenance of achieved results. Think of the medication as a tool that makes healthy choices easier, not a magic solution that works without any effort on your part.
Frequently Asked Questions
How long does it take to reach the maximum Zepbound dose?
Following the standard escalation schedule, reaching the maximum 15 mg dose takes approximately 20 weeks from treatment initiation. This timeline assumes four-week intervals between each dose increase without extended pauses. Some patients remain at lower maintenance doses if they achieve desired results before reaching maximum strength, while others may take longer if they need extended adaptation periods at certain levels. Your journey might be shorter or longer depending on your individual response and goals.
Can I stay at a lower dose if I’m getting good results?
Yes, remaining at a lower dose makes perfect sense when you achieve your weight loss goals and maintain results at that level. Many patients find their optimal maintenance dose falls between 7.5 mg and 12.5 mg rather than requiring maximum strength. Your healthcare provider can help determine whether staying at your current dose or continuing to increase offers the best approach for your specific situation and long-term objectives. There’s no requirement to reach the highest dose if a lower level works well for you.
What happens if I skip a dose increase?
Delaying a scheduled dose increase extends your overall treatment timeline but doesn’t harm the medication’s effectiveness. Valid reasons for postponing an increase include persistent side effects, illness, or major life events that make managing adjustment symptoms difficult. Discuss timing modifications with your provider rather than making changes independently to ensure your approach aligns with your treatment goals. Sometimes staying at a dose longer actually helps you build stronger habits before advancing.
Will I gain weight if I don’t keep increasing the dose?
Weight regain doesn’t automatically occur when you stop increasing doses. Many patients maintain their weight loss at intermediate dose levels for extended periods. The key involves finding your optimal maintenance dose – the level that sustains your results without requiring continuous escalation. Some people need maximum doses for maintenance while others achieve stability at lower concentrations. Your lifestyle habits matter more for long-term maintenance than which dose level you end up using.
How do I know if my dose is too high?
Excessive dosing typically produces persistent, severe side effects that don’t improve after two weeks at the new level. Signs include frequent vomiting, inability to eat adequate nutrition, extreme fatigue, or other symptoms that interfere with daily life. Moderate, temporary side effects during the first week or two after increases represent normal adjustment rather than excessive dosing. Contact your provider if symptoms seem disproportionate to expected adjustment effects or if you can’t function normally in your daily activities.
Can I decrease my dose after reaching a higher level?
Dose reduction is possible when higher levels produce intolerable side effects or when you’ve achieved your weight loss goals and want to find the minimum effective maintenance dose. Some patients successfully maintain their results on lower doses after initially requiring higher concentrations for active weight loss. Any dose adjustments should occur under medical supervision to ensure continued effectiveness and appropriate monitoring. Your provider will help you find the lowest dose that maintains your results.
Does everyone need to reach the maximum 15 mg dose?
No, individual dose requirements vary considerably based on multiple factors including starting weight, metabolic characteristics, and treatment goals. Some patients achieve excellent results at 7.5 mg or 10 mg and never require advancement to maximum strength. The highest dose provides an option for those who need it but doesn’t represent a universal target. Your optimal dose depends on your unique response patterns and objectives rather than a predetermined endpoint that everyone must reach.